This post is about comparing my experiences with fluoxetine (Prozac – an SSRI), psilocybe mushrooms, lion’s mane mushroom, and yerba mate tea. Of course this is my personal experience, not a medical study. Remember that everyone is affected differently by psychoactive compounds. In fact recently my friend told me an interesting scientific theory in regards to why humans differ a lot psychologically. Have you heard of fungi that make ants climb on top of a leaf, hook themselves, and stay there without eating, basically committing ant suicide? The spores of the fungi then burst from the ant and go on to grow into new fungi. Ophiocordyceps unilateralis is called the zombie-ant fungus.
“Researchers think the fungus, found in tropical forests, infects a foraging ant through spores that attach and penetrate the exoskeleton and slowly takes over its behavior.
As the infection advances, the enthralled ant is compelled to leave its nest for a more humid microclimate that’s favorable to the fungus’s growth. The ant is compelled to descend to a vantage point about 10 inches off the ground, sink its jaws into a leaf vein on the north side of a plant, and wait for death.
Meanwhile, the fungus feeds on its victim’s innards until it’s ready for the final stage. Several days after the ant has died, the fungus sends a fruiting body out through the base of the ant’s head, turning its shriveled corpse into a launchpad from which it can jettison its spores and infect new ants.”
So what does this have to do with humans being different? The theory says that humans evolved to react differently to same psychoactive molecules in order to not become victims to simple fungi organisms. Since the infectious fungi are not very complex organisms, they can only release so many molecules. By evolving to have complex brains and having individuals react differently to the same psychoactive molecule, humans became resistant to being overtaken by simple fungi. The theory is that there is no one molecule that a fungi could produce that would make all humans act the same, stop whatever they were doing, walk to a nice moist and wooded area, lie down, and wait for fungi spores to emerge from them.
Back to fluoxetine and shrooms
Fluoxetine
Fluoxetine is a selective serotonin reuptake inhibitor. N-methyl-3-phenyl-3-[4-(trifluoromethyl)phenoxy]propan-1-amine. It delays the reuptake of serotonin, resulting in serotonin persisting longer when it is released. Also dopamine and norepinephrine may contribute to the antidepressant action of fluoxetine in humans.
From wiki: Fluoxetine elicits antidepressant effect by inhibiting serotonin re-uptake in the synapse by binding to the re-uptake pump on the neuronal membrane to increase its availability and enhance neurotransmission. Norfluoxetine and desmethylfluoxetine are metabolites of fluoxetine and also act as serotonin re-uptake inhibitors, so increase the duration of action of the drug. Fluoxetine appeared on the Belgian market in 1986. In the U.S., the FDA gave its final approval in December 1987, and a month later Eli Lilly began marketing Prozac.
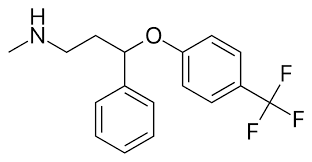
Fluoxetine is one of medications considered to be effective for PMDD (premenstrual dysphoric disorder). Also research indicates that low doses of fluoxetine could help with PMS. PMS appears to be triggered by the fall in secretion of the ovarian sex steroid hormone progesterone that occurs towards the end of the menstrual cycle and leads to a decline in its breakdown product allopregnanolone, which acts in the brain as a potent sedative and tranquilising agent. In other words, women with PMS are undergoing a type of drug withdrawal response from an in-built, tranquilising steroid chemical in their brains. New research shows that antidepressants such as fluoxetine inhibit a specific enzyme in the brain, which deactivates allopregnanolone, therefore maintaining the chemical balance of this in-built tranquiliser in the brain. Recent findings published in the British Journal of Pharmacology, show that short-term treatment with a low dose of fluoxetine immediately prior to the rat’s premenstrual period not only raised brain allopregnanolone and prevented the development of PMS-like symptoms but also blocked the increase in excitability of brain circuits involved in mediating the stress and fear responses that normally occur during this phase of the cycle.
Enzyme identified that could lead to targeted treatment for PMS
A review of studies found that fluoxetine was more tolerabled by female patients than tricyclic amine antidepressants (Amitriptyline, Imipramine). ” In this study, a retrospective analysis of 11 randomized, double-blind, well-controlled trials was done to compare data from 427 female patients on fluoxetine and 423 female patients on TCAs. Both fluoxetine and TCAs significantly reduced the HAMD17 total mean score from baseline to end point, week 5 (fluoxetine, 24.35 to 14.37; TCAs, 24.57 to 14.43; p < 0.001). Both treatment groups were associated with significant reductions in the HAMD17 anxiety/somatization and insomnia subfactor scores. Abnormal vision, constipation, dizziness, dry mouth, and somnolence occurred more frequently (p < 0.05) in the TCA group. Insomnia and nausea were the only adverse events more common (p < 0.05) in the fluoxetine group. This study demonstrates that fluoxetine is an effective and tolerable agent for the treatment of major depressive disorder in women.”
Fluoxetine vs. tricyclic antidepressants in women with major depressive disorder
My experience with fluoxetine – the first time that I took 10mg of fluoxetine, I felt a difference in less than three hours. It was as if I was taken out of a dark basement and into a sunny day in July. Unfortunately I also experienced insomnia that did not go away and I had a sense of apathy, in the end I stopped taking fluoxetine, but I know many women who swear by it.
Psilocybin
Next I will mention psilocybin. Psilocybin is a psychedelic compound produced by more than 200 species of mushrooms. Psilocybin is quickly converted in human body to psilocin. Psilocin is a prtial agonist for several serotonin receptors. An agonist is a chemical that binds to a receptor and activates the receptor to produce a biological response. Recently there has been increased reseach interest in psilocybin and how it could help with depression.
“A landmark study conducted by the Beckley/Imperial Research Programme has provided the first clinical evidence for the efficacy of psilocybin-assisted psychotherapy to treat depression, even in cases where all other treatments have failed. We gave oral psilocybin to 20 patients with treatment-resistant depression, all of whom had previously tried at least two other treatment methods without success. Participants had suffered from depression for an average of 18 years, with severity ranging from moderate to severe. Each patient received two doses of psilocybin (10 and 25mg) 7 days apart, accompanied by psychological support before, during, and after each session. All participants also underwent brain scans to investigate the neural underpinnings of psilocybin mechanisms of action on depression. Follow-up examinations were carried out at 5 weeks, and three and six months. Results highlights All patients showed some reductions in their depression scores at 1-week post-treatment and maximal effects were seen at 5 weeks, with results remaining positive at 3 and 6 months. Notably, reductions in depressive symptoms at 5 weeks were predicted by the quality of the acute psychedelic experience. The drug was also well tolerated by all participants, and no patients sought conventional antidepressant treatment within 5 weeks of the psilocybin intervention. While it is important to note that this was a relatively small study with no control group, placebo, or ‘blinding’ (meaning participants were fully aware what they were getting), the results are extremely encouraging and confirm that psilocybin is safe to give to depressed patients, warranting further research into this area.”
Sceletium tortuosum (Kanna) – a plant commonly found in South Africa. Laboratory studies have found that Sceletium alkaloids are selective serotonin reuptake inhibitors (SSRIs). Thus, they have the same action as pharmaceutical SSRIs such as Prozac. Animal studies have found that Sceletium can improve mood and reduce anxiety-related behaviours.
